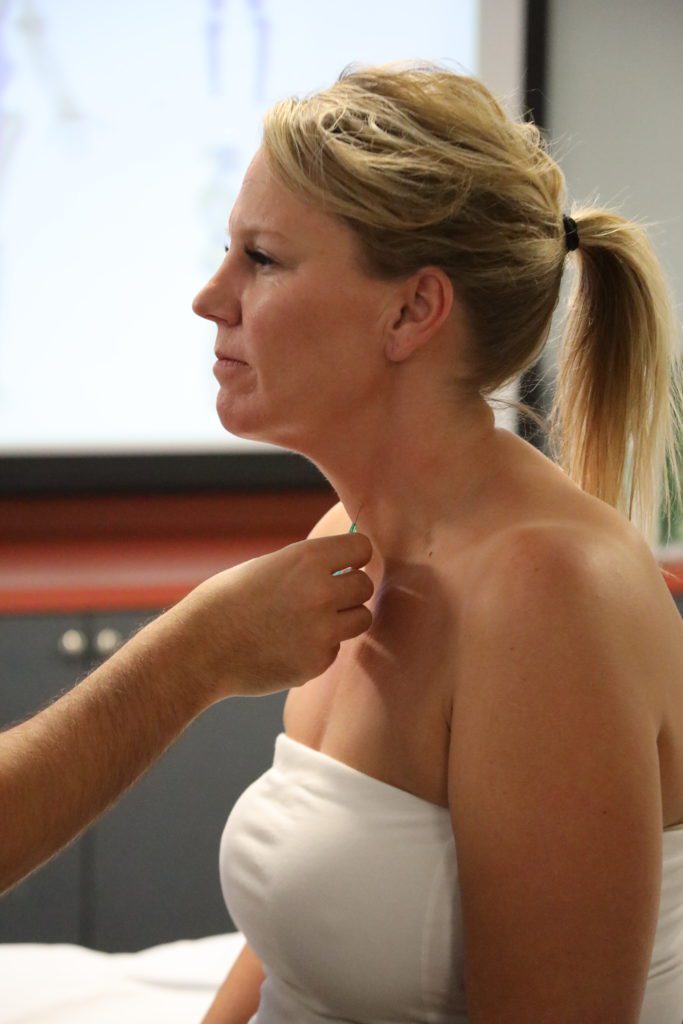
What is Myofascial Pain and Muscle Dysfunction?
Myofascial pain, characterized by the presence of hyper-irritable contractures or taut bands within the soft tissue, featuring trigger points, is one of the leading causes of pain that brings patients to their doctor (Hamid, et al., 2017). Myofascial trigger points oftentimes develop due to overuse, misuse (acute injury), and even underuse, causing continuous contraction within the muscle fibers, giving rise to nociceptor activation, characteristic referred pain, and motor dysfunction. The sustained contraction within muscle fibers leads to ischemia, tissue hypoxia, lowered tissue PH, and ultimately biochemical and histological changes within the trigger point including histamine, serotonin, bradykinin, interleukins, substance P, CGRP, acetylcholine, TNF-a, and cytokines (Dommerholt 2011).
Although muscle has been in the spotlight and subject to several decades of research, the fascia has received an underwhelming amount of attention as a large contributor to the clinical relevance of myofascial pain and musculoskeletal dysfunction. As we graduate from our functional understanding of fascia as a force transmitter and begin to explore the relationships with cellular contraction (myofibroblasts & nerve growth factor), interstitial fluid regulation, and piezoelectricity, the pathological presentations of fascial dysfunction are gaining well-deserved recognition.
How to Treat Myofascial Pain with Fascial Needling
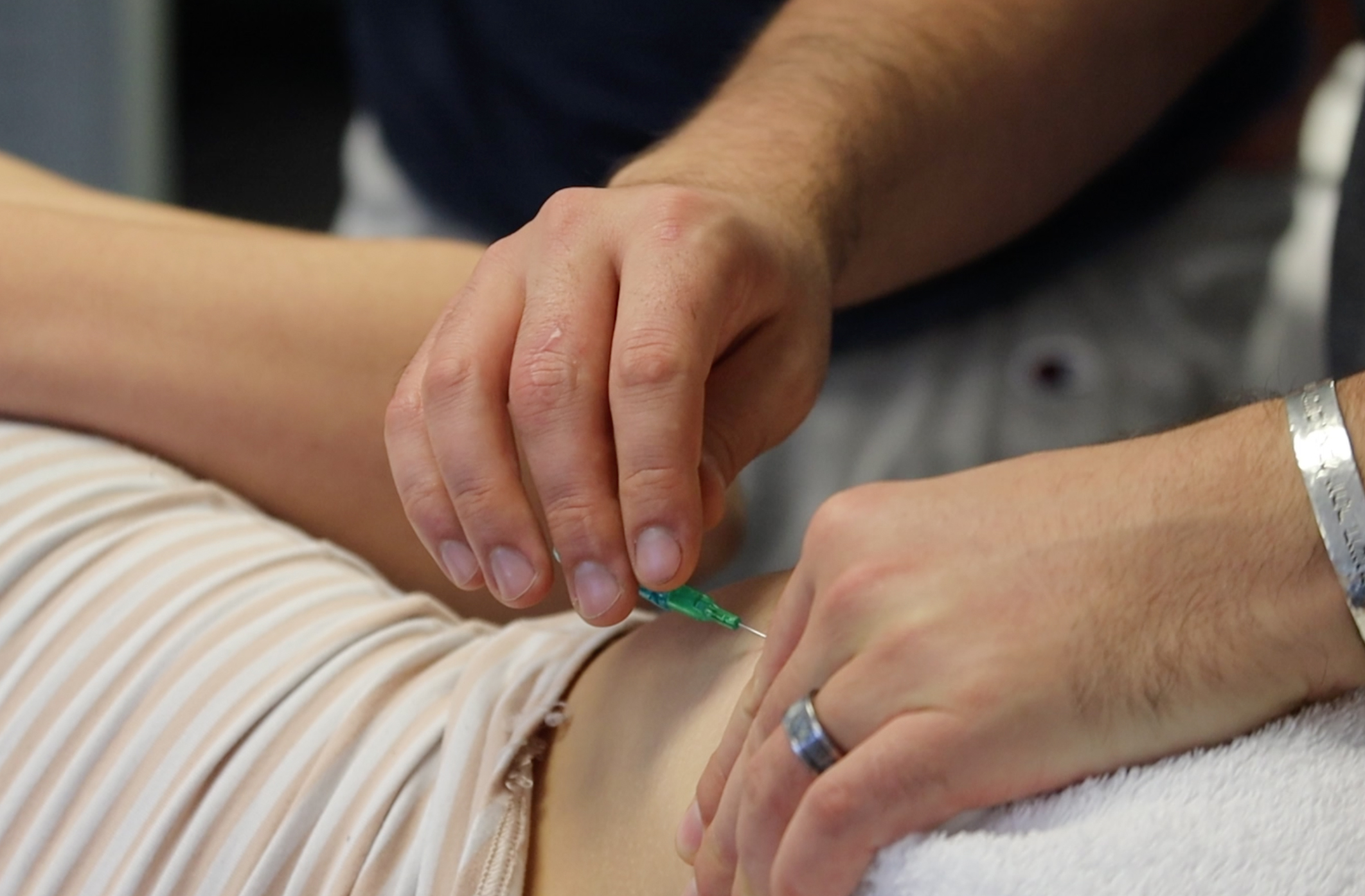
 Fascial needling, specifically FSN-AV (Fu’s Subcutaneous Needling – American Version), is a more recently developed needling therapy to treat myofascial pain and muscle dysfunction. Although the mechanism of action is not fully understood, mechanical stimulation of subcutaneous connective tissue has been shown to induce cytoskeletal remodeling and better alignment of collagen fibers by means of mechanotransduction (Langevin, et al., 2006). The mechanical forces incited within the soft tissue are crucial to the homeostasis of cell and tissue morphology and function as well as influencing the cytoskeletal organization and gene expression (ie. collagen), eventually eliciting a profound effect on the activity of the cell’s nucleus (Kumka and Bonar, 2012). As FSN elicits its effects on the body via the superficial fascia (Fu and Shepherd, 2013) where type I collagen fibers are most abundant, the clinical implications may be profound.
Fascial needling, specifically FSN-AV (Fu’s Subcutaneous Needling – American Version), is a more recently developed needling therapy to treat myofascial pain and muscle dysfunction. Although the mechanism of action is not fully understood, mechanical stimulation of subcutaneous connective tissue has been shown to induce cytoskeletal remodeling and better alignment of collagen fibers by means of mechanotransduction (Langevin, et al., 2006). The mechanical forces incited within the soft tissue are crucial to the homeostasis of cell and tissue morphology and function as well as influencing the cytoskeletal organization and gene expression (ie. collagen), eventually eliciting a profound effect on the activity of the cell’s nucleus (Kumka and Bonar, 2012). As FSN elicits its effects on the body via the superficial fascia (Fu and Shepherd, 2013) where type I collagen fibers are most abundant, the clinical implications may be profound.
As the FSN needle is manipulated within the superficial fascia, there is significant potential to influence mechanoreceptors within the matrix and induce an immediate change in sympathetic tone, realignment of fibroblasts, and changes in viscosity (Schleip, 2003). Fibroblasts, highly sensitive to tensile forces exerted on the fascia, are responsible for synthesis and organization of the ECM (extracellular matrix) and when stretched respond by producing more collagen and proteoglycans such as GAGs (glycosaminoglycans) and the non-sulfated (HA) hyaluronic acid. Proteoglycans are the main regulators of tissue densification and thus viscoelastic behavior within the matrix, due to their negative charge and ability to attract water to produce changes within the ECM, indirectly determining the function of the different systems dealing with the fascial continuum (Langevin, 2004). Fibroblasts within areolar connective tissue exhibit dynamic cytoskeletal remodeling within minutes in response to tissue stretch ex vivo and in vivo (Langevin, et al., 2011). On the contrary, other studies have shown that a longer duration is necessary for permanent viscoelastic deformation of fascia to be achieved (Schleip, 2003).
Furthermore, actin containing smooth muscle cells known as myofibroblasts have been identified in recent research to be contained within the fascia, working in tandem with the GAGs in the ECM to control tissue densification and contraction of the fascia (Schleip, et al., 2018), which Tom Myers described as cellular tensegrity. Sympathetic activation increases cytokine expression in the body, which has a stimulatory effect on myofibroblast contraction, thereby leading to an increase in fascial stiffness. Fascial stiffness can be further dictated by fibrosis of fascia due to trauma, surgery, diabetes, or age-related degeneration (Pavan, et al., 2014).
Causes and Impact of Fascial Stiffness
Resting muscle tone may be significantly influenced by changes in fascial stiffness (Masi and Hannon, 2008), but tension in myofascial tissue is also actively regulated by myofibroblasts with the potential to impact active musculoskeletal dynamics (Schleip, et al., 2019). The fascia and underlying muscle can influence one another via their intimate mechanical interaction between the contractile element and the muscle-related fascial structures. Within the muscle belly, the actin and myosin filaments and the ECM within the fascia are interconnected (Schleip, et al., 2018). Furthermore, all three layers of intramuscular fascia (endo/peri/epimysium) are continuous with collagenous structures receiving direct influence from the superficial fascia. To assess the thickness of the membranous layer of the superficial fascia in various regions of the body, a dissection of six cadavers along with ultrasound imaging of four subjects was performed.
In all six cadavers, a continuous layer of fibrous membrane in the superficial fascia was found consistently in all the dissected regions of the body and was also confirmed by ultrasonography (Abu-Hijleh, et al., 2006).
One study concluded the connective tissue and antagonistic muscles of the lower limb (anterior tibial compartment and the peroneal compartment) were tightly fused (Huijing, et al., 2007), along with another study concluding that a fascial attachment was detected between the semitendinosus and the gastrocnemius (Tuncay, et al., 2007). Furthermore, there is a direct insert of the deltoid, latissimus dorsi, and pectoralis minor into the brachial fascia (Stecco, et al., 2016).
Other Causes of Myofascial Pain
Although myofascial pain is characterized by the presence of muscular trigger points and muscle dysfunction, we know that the myofascial system can carry neural signals and any alteration in the connective tissue structure can alter the polarization of muscle fibers, leading to spontaneous muscle contraction (Bordoni, et al., 2020). Change to the GAGs leading to increased viscosity within the ECM will cause the fascia to have difficulty sliding between layers, compromising both the deep fascia and the underlying muscle, leading to the development of myofascial pain (Stecco, et al, 2013).
Fascial Needling Results
The piezoelectric nature of collagen-rich tissues has been known for some time. With FSN’s strong influence on collagen fibers, the swaying movement of the therapy when performed within the superficial fascia can elicit a piezoelectric effect, improving cell ion channels, diffusing and transmitting the effect to distant muscles (Huang, et al., 2020), eliciting an immediate change in tissue plasticity.
As each muscle fiber is physically encased in an aponeurotic fascia and on a cellular level the passive properties of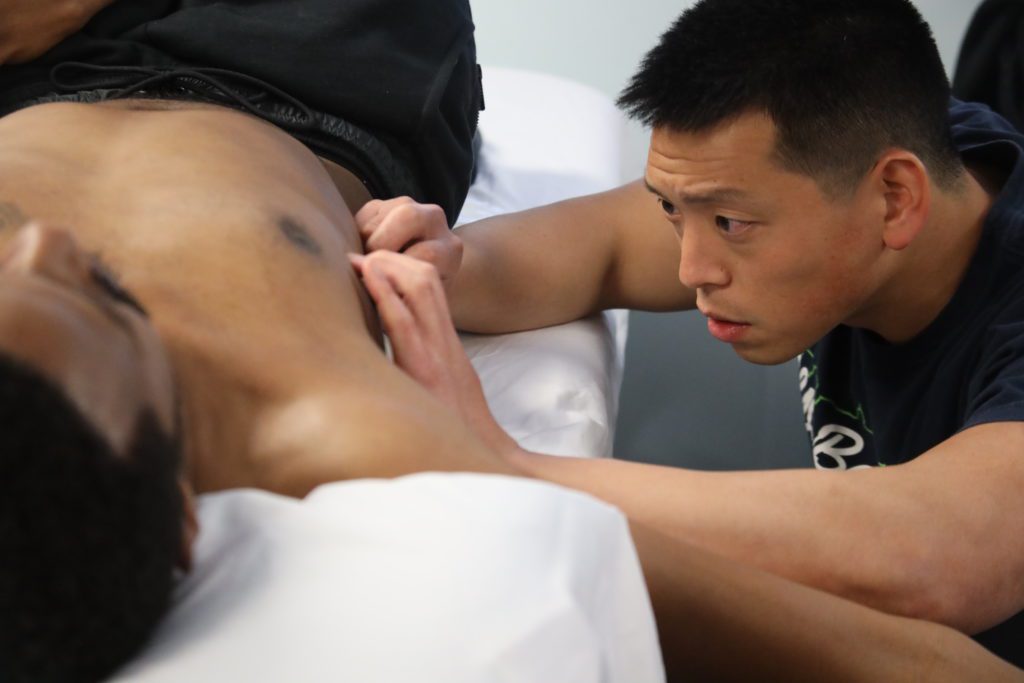 muscle are dependent on viscoelasticity by interacting directly with the ECM, it may be possible to further extrapolate that a reduction in tissue stiffness can increase oxygenation and therefore increase tissue PH, increasing acetylcholinesterase activity and reducing the concentration of nociceptive substances within the muscle. Manipulation of the superficial fascia using a fascial needle has been shown to elicit changes in the density of the ECM and while more research has to be carried out, there is a possibility that due to the intimate relationship between the connective tissue and muscle, a fascial needling therapy such as FSN can elicit change in the biochemical milieu of myofascial trigger points.
muscle are dependent on viscoelasticity by interacting directly with the ECM, it may be possible to further extrapolate that a reduction in tissue stiffness can increase oxygenation and therefore increase tissue PH, increasing acetylcholinesterase activity and reducing the concentration of nociceptive substances within the muscle. Manipulation of the superficial fascia using a fascial needle has been shown to elicit changes in the density of the ECM and while more research has to be carried out, there is a possibility that due to the intimate relationship between the connective tissue and muscle, a fascial needling therapy such as FSN can elicit change in the biochemical milieu of myofascial trigger points.
Ryan Shepherd, ND, AP, LAc, CMTPT
References
Abu Hijeh, M.F., Roshier, A.L., Al-Shboul, Q., Dharap, A.S., Harris, P.F. (2006). The membranous layer of superficial fascia: evidence for its widespread distribution in the body. Surgical and Radiological Anatomy. 28. 606-619.
Bordoni, B., Zanier, E. (2014). Clinical and symptomatological reflections: The Fascial System. Journal of Multidisciplinary Healthcare. 7. 401-411.
Bordoni, B., Myers, T. (2020). A Review of the Theoretical Fascial Models: Biotensegrity, Fascintegrity, and Myofascial Chains. Cureus. 12(2), e7092.
Cowman, M.K., Schmidt, T.A., Raghavan, P., Stecco. (2015). Viscoelastic Properties of Hyaluronan in Physiological Conditions. F100Res. 4. 622.
Dommerholt, J, 2011. Dry needling — peripheral and central considerations. J Manual Manipul Ther, 19, 223-237.
Hamid, T.N., Mehrdad, N., Jafar, S., Taghi, J.M., Massoud, A.A., Ramin Y. (2017). Histology and Biochemical Milieu of Trigger Points: A Literature Review. Asian Journal of Health and Medical Research. 3(1), 3-9.
Huang, H., Liu, J., Fu, M., Lin, I.W., Chou, L.W. (2020). Fu’s subcutaneous needling for subcutaneous adhesions and scar hyperplasia in the neck region. Medicine (Baltimore). 99(29).
Huijing, P.A., Van de Langenberg, R.W., Meesters, J.J., Baan, G.C. (2007). Extramuscular myofascial force transmission also occurs between synergistic muscles and antagonistic muscles. Journal of Electromyography and Kinesiology 17(6). 680-690.
Kumka, M., Bonar, J. (2012). Fascia: a morphological description and classification system based on a literature review. The Journal of the Canadian Chiropractic Association.
Langevin, H.M., Bouffard, N.A., Badger, G.J., Latridis, J.C., Howe, A.K. (2005). Dynamic fibroblast cytoskeletal response to subcutaneous tissue stretch ex vivo and in vivo. American. Physiological Society. 288(3).
Langevin, H.M., Bouffard, N.A., Badger, G.J., Churchill, D.L., Howe, A.K. (2006). Subcutaneous tissue fibroblast cytoskeletal remodeling induced by acupuncture: Evidence for a mechanotransduction‐based mechanism. Journal of Cellular Physiology.
Masi, A.T., Hannon, J.C. (2008). Human resting muscle tone (HRMT): narrative introduction and modern concepts. Journal of Bodywork and Movement Therapies. 13(2). 118-120.
Schleip, R. (2003). Fascial plasticity — a new neurobiological explanation. Journal of Bodywork and Movement Therapies. 7(1), 11-19.
Schleip, R., Yucesoy, C.A., Banzer, W. (2018). Not merely a protective packing organ? A review of fascia and its force transmission capacity. Journal of Applied Physiology. 124(1).
Schleip, R., (2019). Fascia Is Able to Actively Contract and May Thereby Influence Musculoskeletal Dynamics: A Histochemical and Mechanographic Investigation. Frontiers in Physiology. 10.335
Stecco, C., Porzionato, A., Macchi, V., Stecco, A., Vigato, E., Parenti. A., Delmas, V., Aldegheri, R., De Caro, R. (2008). The Expansion of the Pectoral Girdle Muscles onto the Brachial Fascia: Morphological Aspects and Spatial Disposition. Karger. 188. 320-329.
Tuncay, I., Kucuker, H., Uzun, I., Karalezli, N. (2007). The fascial band from semitendinosus to gastrocnemius: the critical point of hamstring harvesting: an anatomical study of 23 cadavers. Acta Orthopaedica. 78(3).
Zullo, A., Fleckenstein, J., Schleip, R., Hoppe, K., Wearing, S., Klinger, W. (2020). Structural and Functional Changes in the Coupling of Fascial Tissue, Skeletal Muscle, and Nerves During Aging.