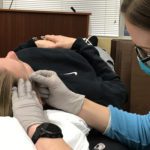
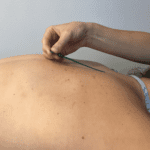
Antithrombotic Drugs and Dry Needling: Uncharted Territory?
Although dry needling has been used throughout the world for decades, the question of whether dry needling is safe for patients taking antithrombotic drugs has not been explored, leaving clinicians and patients in a vacuum. A recent review article, co-authored by Jan Dommerholt, addresses this issue and provides guidance on whether dry needling can be used safely in patients with altered coagulation (1). According to the American Physical Therapy Association (APTA), “patients with an abnormal bleeding tendency must be needled with caution. Dry needling of deep muscles that cannot be approached with direct pressure to create hemostasis may need to be avoided to prevent excessive bleeding” (2), but the APTA did not cite any source document, simply because there were no such references at the time of publication.

Of course, the objective of dry needling is not to target blood vessels, but since they are present anywhere in the body, it is impossible to get around them. Not surprisingly, in an Irish adverse events study (also co-authored by Jan Dommerholt), graduates of our dry needling courses reported that minor bleeding was the most common adverse event (3). Whether the 36 continuing education companies we counted recently in the US alone have similar or different risk profiles, is not known. A similar study conducted in the US confirmed that minor bleeding may occur with dry needling (4). The risk of bleeding may depend on the kind of dry needling, the diameter and characteristics of the needle, the body region, and the level of education and skill of the practitioner. Other factors may include the patient’s age, gender, renal function, and possible food and drug interactions.
A Closer Look at Antithrombotic Medications
It seems fair to assume that not all dry needling practitioners have a solid working knowledge of antithrombotic medications. Let’s take a closer look. There are two classes of antithrombotic drugs: antiplatelet agents and anticoagulants. Aspirin and P2Y12 receptor blockers are the main antiplatelet agents. Anticoagulants include vitamin K antagonists (VKAs) and direct oral anticoagulants (DOACs), and heparins, which are administered subcutaneously or intravenously (Figure 1).

Antiplatelet Agents
Aspirin, P2Y12 Receptor Blockers, and Dual Antiplatelet Therapy
The first-line antiplatelet medication is aspirin, which is used in patients with angina, coronary artery stents, previous heart attack or stroke, after coronary artery bypass graft surgery, and in patients undergoing surgery for hip fracture, to prevent the formation of blood clots. P2Y12 receptor blockers, such as clopidogrel, prasugrel, and ticagrelor, are often used in combination with aspirin, which is referred to as dual antiplatelet therapy (DAPT). DAPT is used primarily to prevent thrombosis following angioplasty with stent implantation or balloon angioplasty even though combining two antiplatelet medications increases the risk of bleeding.
Anticoagulants
Vitamin K Antagonists (VKAs)
The vitamin K antagonist warfarin is used most in the US, whereas in Europe phenprocoumon and acenocoumarol are more widely prescribed to prevent and treat venous thromboembolism, systemic embolism, or stroke in patients with prosthetic heart valves or atrial fibrillation. Patients who are taking VKAs must be monitored closely. A key factor is the International Normalized Ratio (INR), which is a standardized method for reporting results of the prothrombin time (PT assay). Patients with a stable INR have a lower risk of bleeding. An INR range of 2.0–3.0 is considered the therapeutic range and recommended by international guidelines. The US Food and Drug Administration (FDA) has approved the antidote 4-Factor Prothrombin Complex Concentrate (PCC) specific for VKAs.
Direct Oral Anticoagulants (DOACs)
It is likely that DOACs, which typically have more favorable therapeutic profiles, will replace VKAs over time. They are more predictable and require less therapeutic monitoring, but they still have certain drug-drug interactions. DOACs are associated with significant reductions in intracranial hemorrhage, stroke, and mortality. The FDA has approved five DOACs: dabigatran, rivaroxaban, apixaban, edoxaban, and betrixaban. FDA-approved antidotes for DOACs are idarucizumab for dabigatran, and andexanet alfa for rivaroxaban and apixaban. PCC may be used off-label for reversing DOACs.
Heparins (Subcutaneous and Intravenous)
Heparins are prescribed, often combined with DAPT in patients with heart failure or prolonged immobilization in bed or in patients with acute coronary syndrome. They reduce complications of venous thromboembolism in high-risk medical conditions. There are two types of heparins: unfractionated heparins (UFH), which are administered subcutaneously or intravenously, and low molecular weight heparins (LMWH), which are administered subcutaneously. The LMWH has gradually replaced UFH for most clinical indications.
Relationship Between Antithrombotics and Dry Needling
Because there are no studies of the risk of bleeding after dry needling, we evaluated other common needling procedures such as electromyography, acupuncture, botulinum toxin injections, and ultrasound-guided fine-needle aspiration biopsies, realizing the differences in applications and types of needles (1). For the details of the analysis, please review the published paper. In summary, if specific dry needling and individual risks are properly considered, antithrombotic medications do not change the risk and safety profile of dry needling and should not be considered an absolute contraindication for dry needling. To improve the safety of DN practice in patients taking antithrombotics, we recommend using ultrasound guidance, especially for dry needling close to major blood vessels or in deeper muscles where hemostasis cannot be applied. For nonanticoagulated patients, it is common to apply pressure for about 5 seconds. For patients taking anticoagulants, the pressure should be applied for about 10–15 seconds.
Learn From the Best
Dr. Jan Dommerholt is co-founder, president, and CEO of Myopain Seminars. In 1995-1997, Dr. Dommerholt introduced dry needling to the United States and together with Robert Gerwin he started teaching Manual Trigger Point Therapy courses. He has taught hundreds of courses and lectured at over 80 conferences throughout the United States, Canada, Mexico, South America, Europe, Australia, and the Middle East while maintaining an active clinical practice.

References
- Muñoz M, Dommerholt J, Pérez-Palomares S, Herrero P, Calvo S. Dry Needling and Antithrombotic Drugs. Pain Research and Management, vol. 2022, Article ID 1363477.
- American Physical Therapy Association, Description of Dry Needling in Clinical Practice: An Educational Resource Paper, American Physical Therapy Association, Alexandria, VA, USA, 2013.
- Brady S, McEvoy J, Dommerholt J, Doody C. Adverse events following dry needling: A prospective survey of Chartered Physiotherapists. J Manual Manipul Ther. 2014;22(3):134-140.
- Boyce D, Wempe H, Campbell C, et al. Adverse events associated with therapeutic dry needling. Intern J Sports Phys Ther. 2020;15(1):103-113.